
Much of the work carried out by DTT is in support of the National Toxicology Program (NTP), an interagency partnership of the Food and Drug Administration, National Institute for Occupational Safety and Health, and NIEHS.
The Comparative and Molecular Pathogenesis Branch (CMPB) provides toxicologic pathology leadership and laboratory animal medicine expertise to meet the goals of the NIEHS Division of Translational Toxicology (DTT) using new technologies in biomedical research. DTT is a team-based scientific organization, and its mission is to improve public health through development of data and knowledge that are translatable, predictive, and timely.
A major function of the Branch is to provide a translational and mechanistic context to pathology outcomes with the integration of scientific data across all DTT branches and offices. In addition, CMPB trains pathology and laboratory animal medicine fellows to be leaders in multidisciplinary research.
CMPB scientists and staff are responsible for the following general functions:
- Developing and sharing educational and training materials that enhance the understanding and practice of toxicologic pathology with the international community through the publicly available online DTT Global Toxicologic Pathology Training Program.
- Developing new methods, approaches, and technologies in pathology and laboratory animal medicine.
- Directing, managing, evaluating, and interpreting all pathology data generated during the conduct of DTT toxicity and carcinogenicity studies.
- Directing the DTT laboratory animal medicine program.
- Establishing standards, terminology, and diagnostic criteria for rodent pathology (NTP Nonneoplastic Lesion Atlas).
- Investigating molecular mechanisms of spontaneous and chemically induced nonneoplastic and neoplastic lesions in DTT studies.
- Leading state-of the-science core pathology laboratories which include:
- Clinical chemistry
- Electron Microscopy
- Hematology
- Histology
- Imaging Sciences/Artificial Intelligence
- Immunohistochemistry
- Laser Capture Microdissection
- Mouse Embryo Phenotyping
- Necropsies
- Overseeing the scientific activities of the DTT archives and frozen tissue bank.
- Providing diagnostic pathology expertise and collaborations with NIEHS investigators and the NIEHS laboratory animal colony.
CMPB is organized into eight scientific groups that are led by board certified pathologists and laboratory animal medicine scientists, and fellows of the International Academy of Toxicologic Pathology.
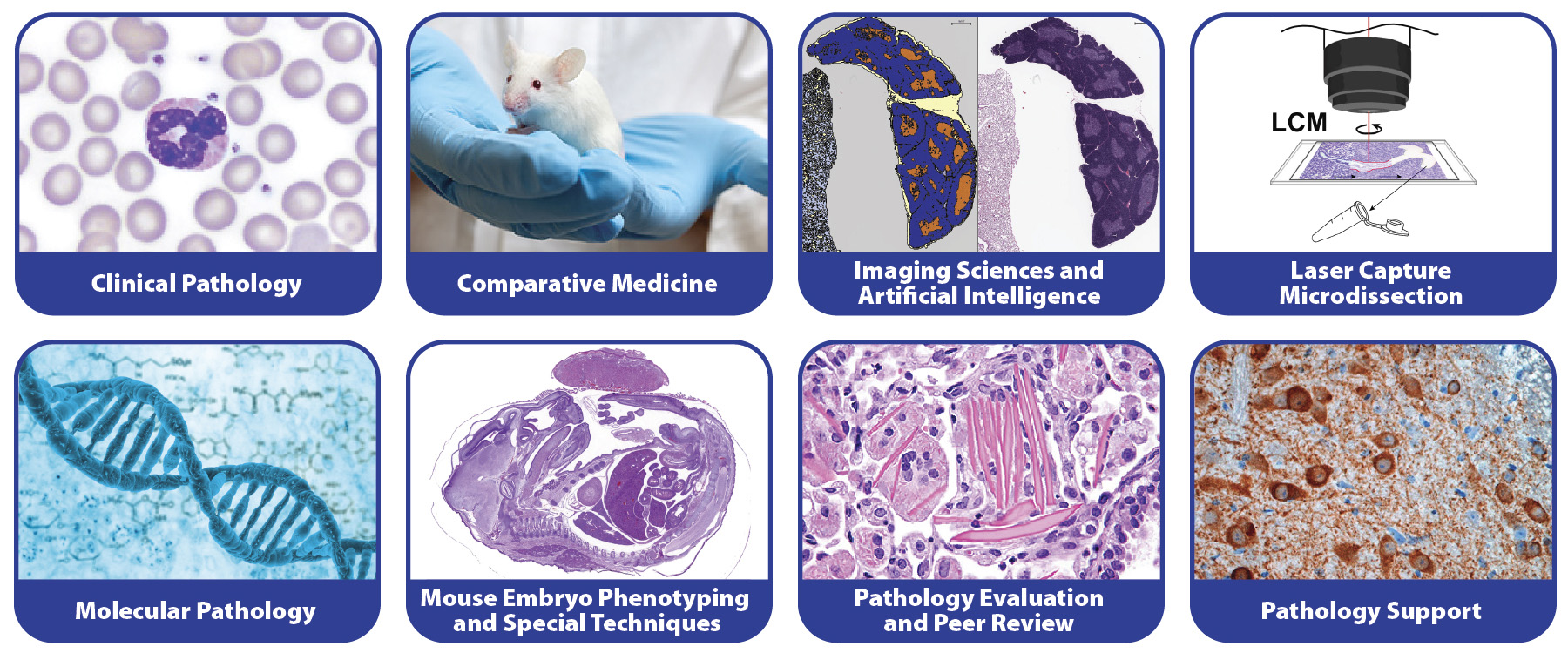
- Clinical Pathology Group
- Comparative Medicine Group
- Imaging Sciences and Artificial Intelligence Group
- Laser Capture Microdissection Group
- Molecular Pathology Group
- Mouse Embryo Phenotyping and Special Techniques Group
- Pathology Evaluation and Peer Review Group
- Pathology Support Group
CMPB scientists contribute to NTP report and monograph series and publish findings in the NIEHS report series and peer-reviewed scientific literature. Staff publications can be viewed on the webpage.
Chief

Robert C. Sills, D.V.M., Ph.D.
[email protected]


