Introduction
Aloe is a succulent plant widely used in alternative medicine. There are at least 420 different plant species of Aloe. Aloe vera specifically refers to the Aloe barbadensis Miller plant, which is the most common form used in Aloe-based products.
How is Aloe used?
Traditionally, the clear gel from the Aloe plant is rubbed on the skin as an ointment to treat wounds and burns. The green part of the leaf can be made into a juice or dried and taken orally as a laxative. Aloe vera is used in many commercial products in various forms, including drinks, concentrates, capsules, powders, and as a flavoring.
What if I use Aloe vera gel on my skin, will it cause me harm?
Applying Aloe vera gel on the skin is not likely to cause harm. Although these particular NTP studies only looked at oral exposure to Aloe vera, other NTP studies examined the effects of the gel on the skin and did not find a strong link between Aloe vera in skin care products, sunlight, and the development of skin cancer. The results of those skin studies were reported in the NTP Technical Report 553 on the Photococarcinogenesis Study of Aloe Vera.
Why would people consume drinks that have Aloe in them?
Product advertisements claim the drinks can be used to cleanse the digestive system or relieve constipation.
What makes Aloe act like a laxative?
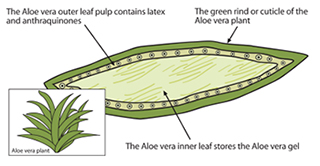
The outer pulp of Aloe leaves, known as the latex, contains anthraquinones. Aloin is an anthraquinone found in the latex that may give the plant its laxative qualities.
How much aloin is in products in the U.S. that may be consumed by the public?
There are very little data about what the levels of aloin are in the consumer products. According to published research, levels of aloin in most liquid products are less than 1 part per million (ppm); however, levels in solid or semi-solid products may be 10-100 times higher.
How much aloin is in a typical drink?
This is not clear. Industry usually processes the Aloe vera leaf to make a decolorized whole leaf extract to remove aloin. Industry has a self-regulated upper limit standard of 10 ppm of aloin in orally ingested products, but there are no labeling requirements for aloin content.
What are the adverse effects of excessive Aloe exposure in humans?
More research is needed to answer this question. In rodents, it caused intestinal tumors. If you are concerned, have a medical condition, or are taking other drugs, herbs, or supplements, you should speak with your physician before altering existing treatments or starting any new treatment.
How similar are the doses of aloin in the drinking water fed to the animals to those found in Aloe vera products?
This is not clear, as there are no labeling requirements to list aloin content. The concentration of aloin in the water that the rodents drank in the NTP study is similar to that found in some Aloe vera products, but higher than in other products. The NTP wanted to test an Aloe preparation that includes all components that may be in the products on the market. The NTP study used a whole leaf extract of Aloe barbadensis Miller that did not undergo charcoal filtration during processing. Many commercial producers of Aloe vera beverages use charcoal filtration to reduce the levels of aloin in their products.
Is Aloe vera regulated?
Aloe vera is a dietary supplement and not a regulated drug. There is no guarantee of strength, purity, or safety of these products. Aloe vera is approved by the Food and Drug Administration as a food additive for flavor.
What do the NTP rodent studies mean for humans?
The NTP rat studies give cause for serious concern. Some questions that need to be answered before their importance can be fully appreciated include:
- What products are actually in the market place?
- What are the aloin levels across a broad spectrum of products?
- What are the patterns of human exposure?
There is not enough information currently available to know how much Aloe vera people are consuming or for how long. Based on what we know right now, there is nothing that would lead us to believe that this finding would not be relevant to humans.
What could cause the tumors and other effects?
We suspect the aloin but are not sure. We hope to do more studies to determine what may be causing the tumors.
Should I stop drinking Aloe?
That is a decision you might want to make with your health care provider. Like any personal decision, the more information you have the better. Currently, manufacturers of these products are not required to label aloin concentration in Aloe vera products. If aloin is the cause of the tumors in rats then, generally speaking, the lower the concentration of aloin in a product, the lower the possibility that it may cause harm. Tumors developed in animals that drank water containing 60 ppm of aloin daily for nearly their entire lifetime.
Further Reading
Additional Resources
- Botanical Safety Consortium — This consortium, which includes NIEHS and FDA, is a forum for scientists to integrate existing safety data and toxicology tools to evaluate botanical safety
- National Center for Complementary and Integrative Health NCCIH resources on Aloe Vera
- NTP Technical Report 577 on the Toxicology and Carcinogenesis Studies of a Non-decolorized Whole Leaf Extract of Aloe Barbadensis Miller (Aloe Vera) in F344/N Rats and B6C3F Mice (Drinking Water Study)